what cellular structure is selectively permeable for specific items to freely pass through it
Chapter 3: Introduction to Cell Structure and Function
3.5 Passive Transport
Past the end of this section, you lot will be able to:
- Explain why and how passive transport occurs
- Understand the processes of osmosis and diffusion
- Define tonicity and describe its relevance to passive ship
Plasma membranes must allow certain substances to enter and leave a jail cell, while preventing harmful cloth from entering and essential material from leaving. In other words, plasma membranes are selectively permeable—they allow some substances through but not others. If they were to lose this selectivity, the jail cell would no longer be able to sustain itself, and it would be destroyed. Some cells require larger amounts of specific substances than do other cells; they must take a way of obtaining these materials from the extracellular fluids. This may happen passively, as sure materials move back and forth, or the cell may accept special mechanisms that ensure transport. Nigh cells expend most of their energy, in the form of adenosine triphosphate (ATP), to create and maintain an uneven distribution of ions on the opposite sides of their membranes. The structure of the plasma membrane contributes to these functions, but it also presents some problems.
The most straight forms of membrane transport are passive. Passive transport is a naturally occurring phenomenon and does not require the prison cell to expend energy to reach the movement. In passive transport, substances movement from an area of higher concentration to an expanse of lower concentration in a process called diffusion. A physical space in which there is a dissimilar concentration of a unmarried substance is said to take a concentration gradient.
Selective Permeability
Plasma membranes are asymmetric, pregnant that despite the mirror image formed by the phospholipids, the interior of the membrane is non identical to the outside of the membrane. Integral proteins that human activity as channels or pumps work in one direction. Carbohydrates, attached to lipids or proteins, are also plant on the exterior surface of the plasma membrane. These carbohydrate complexes assist the prison cell bind substances that the cell needs in the extracellular fluid. This adds considerably to the selective nature of plasma membranes.
Recall that plasma membranes take hydrophilic and hydrophobic regions. This characteristic helps the movement of certain materials through the membrane and hinders the movement of others. Lipid-soluble textile can hands skid through the hydrophobic lipid core of the membrane. Substances such as the fat-soluble vitamins A, D, E, and One thousand readily pass through the plasma membranes in the digestive tract and other tissues. Fat-soluble drugs also gain easy entry into cells and are readily transported into the body'southward tissues and organs. Molecules of oxygen and carbon dioxide have no charge and pass through by simple improvidence.
Polar substances, with the exception of h2o, present issues for the membrane. While some polar molecules connect easily with the outside of a cell, they cannot readily laissez passer through the lipid cadre of the plasma membrane. Additionally, whereas small ions could hands slip through the spaces in the mosaic of the membrane, their charge prevents them from doing and then. Ions such as sodium, potassium, calcium, and chloride must have a special means of penetrating plasma membranes. Unproblematic sugars and amino acids also demand help with transport across plasma membranes.
Diffusion
Diffusion is a passive process of send. A unmarried substance tends to move from an area of high concentration to an area of depression concentration until the concentration is equal across the space. Y'all are familiar with diffusion of substances through the air. For case, retrieve about someone opening a canteen of perfume in a room filled with people. The perfume is at its highest concentration in the canteen and is at its lowest at the edges of the room. The perfume vapor will diffuse, or spread away, from the canteen, and gradually, more and more people volition scent the perfume as it spreads. Materials motion within the cell's cytosol by diffusion, and sure materials move through the plasma membrane by diffusion (Figure 3.24). Diffusion expends no energy. Rather the unlike concentrations of materials in dissimilar areas are a form of potential energy, and improvidence is the dissipation of that potential energy as materials move down their concentration gradients, from loftier to low.
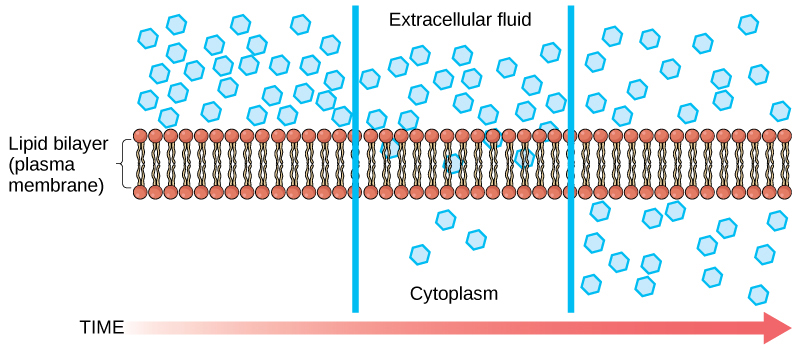
Each separate substance in a medium, such as the extracellular fluid, has its own concentration slope, independent of the concentration gradients of other materials. Additionally, each substance volition diffuse according to that gradient.
Several factors bear on the rate of improvidence.
- Extent of the concentration gradient: The greater the departure in concentration, the more rapid the improvidence. The closer the distribution of the cloth gets to equilibrium, the slower the charge per unit of diffusion becomes.
- Mass of the molecules diffusing: More massive molecules move more than slowly, because information technology is more difficult for them to move between the molecules of the substance they are moving through; therefore, they diffuse more slowly.
- Temperature: College temperatures increase the energy and therefore the movement of the molecules, increasing the rate of diffusion.
- Solvent density: As the density of the solvent increases, the rate of diffusion decreases. The molecules wearisome down because they have a more difficult time getting through the denser medium.
Concept in Action
For an animation of the diffusion process in activity, view this short video on cell membrane transport.

Facilitated transport
In facilitated transport, besides called facilitated diffusion, material moves across the plasma membrane with the aid of transmembrane proteins downward a concentration gradient (from high to depression concentration) without the expenditure of cellular energy. However, the substances that undergo facilitated transport would otherwise not lengthened easily or apace across the plasma membrane. The solution to moving polar substances and other substances across the plasma membrane rests in the proteins that span its surface. The cloth being transported is first fastened to poly peptide or glycoprotein receptors on the exterior surface of the plasma membrane. This allows the material that is needed past the jail cell to be removed from the extracellular fluid. The substances are then passed to specific integral proteins that facilitate their passage, because they class channels or pores that allow certain substances to laissez passer through the membrane. The integral proteins involved in facilitated transport are collectively referred to equally transport proteins, and they function as either channels for the material or carriers.
Osmosis
Osmosis is the diffusion of water through a semipermeable membrane according to the concentration gradient of h2o across the membrane. Whereas improvidence transports material beyond membranes and inside cells, osmosis transports only water across a membrane and the membrane limits the improvidence of solutes in the water. Osmosis is a special case of diffusion. Water, like other substances, moves from an area of higher concentration to one of lower concentration. Imagine a beaker with a semipermeable membrane, separating the two sides or halves (Figure iii.25). On both sides of the membrane, the water level is the aforementioned, simply there are different concentrations on each side of a dissolved substance, or solute, that cannot cross the membrane. If the book of the water is the aforementioned, but the concentrations of solute are different, so there are also unlike concentrations of h2o, the solvent, on either side of the membrane.
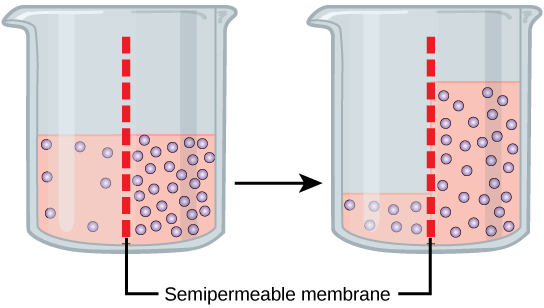
A principle of diffusion is that the molecules move effectually and volition spread evenly throughout the medium if they tin can. Nevertheless, only the material capable of getting through the membrane will diffuse through it. In this case, the solute cannot diffuse through the membrane, but the water tin can. H2o has a concentration gradient in this system. Therefore, water will lengthened down its concentration gradient, crossing the membrane to the side where it is less concentrated. This diffusion of water through the membrane—osmosis—will continue until the concentration gradient of water goes to zero. Osmosis gain constantly in living systems.
Tonicity
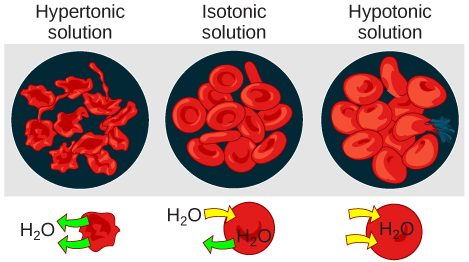
Tonicity describes the amount of solute in a solution. The mensurate of the tonicity of a solution, or the total amount of solutes dissolved in a specific amount of solution, is called its osmolarity. Three terms—hypotonic, isotonic, and hypertonic—are used to relate the osmolarity of a cell to the osmolarity of the extracellular fluid that contains the cells. In a hypotonic solution, such every bit tap water, the extracellular fluid has a lower concentration of solutes than the fluid within the cell, and h2o enters the cell. (In living systems, the point of reference is always the cytoplasm, and so the prefix hypo– means that the extracellular fluid has a lower concentration of solutes, or a lower osmolarity, than the jail cell cytoplasm.) Information technology also means that the extracellular fluid has a higher concentration of water than does the prison cell. In this situation, water will follow its concentration gradient and enter the cell. This may crusade an animal cell to flare-up, or lyse.
In a hypertonic solution (the prefix hyper– refers to the extracellular fluid having a higher concentration of solutes than the cell's cytoplasm), the fluid contains less water than the cell does, such as seawater. Because the jail cell has a lower concentration of solutes, the water volition leave the cell. In outcome, the solute is drawing the h2o out of the cell. This may crusade an fauna prison cell to shrivel, or crenate.
In an isotonic solution, the extracellular fluid has the aforementioned osmolarity every bit the cell. If the concentration of solutes of the prison cell matches that of the extracellular fluid, there will exist no net motility of water into or out of the cell. Claret cells in hypertonic, isotonic, and hypotonic solutions accept on feature appearances (Effigy 3.26).
A doctor injects a patient with what the doctor thinks is isotonic saline solution. The patient dies, and autopsy reveals that many red claret cells accept been destroyed. Do you lot remember the solution the medico injected was actually isotonic?
<!– No, it must take been hypotonic, as a hypotonic solution would cause water to enter the cells, thereby making them burst. –>
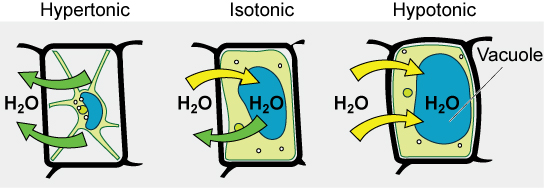
Some organisms, such every bit plants, fungi, leaner, and some protists, accept cell walls that surround the plasma membrane and prevent jail cell lysis. The plasma membrane can only expand to the limit of the cell wall, so the cell will not lyse. In fact, the cytoplasm in plants is always slightly hypertonic compared to the cellular surround, and water will always enter a jail cell if h2o is available. This influx of h2o produces turgor pressure, which stiffens the cell walls of the plant (Figure 3.27). In nonwoody plants, turgor pressure supports the plant. If the plant cells become hypertonic, as occurs in drought or if a plant is not watered fairly, h2o will leave the cell. Plants lose turgor pressure in this condition and wilt.
Department Summary
The passive forms of transport, diffusion and osmosis, move textile of small molecular weight. Substances diffuse from areas of loftier concentration to areas of depression concentration, and this procedure continues until the substance is evenly distributed in a system. In solutions of more than one substance, each type of molecule diffuses co-ordinate to its own concentration slope. Many factors can affect the rate of diffusion, including concentration gradient, the sizes of the particles that are diffusing, and the temperature of the system.
In living systems, diffusion of substances into and out of cells is mediated by the plasma membrane. Some materials lengthened readily through the membrane, but others are hindered, and their passage is only made possible by protein channels and carriers. The chemistry of living things occurs in aqueous solutions, and balancing the concentrations of those solutions is an ongoing trouble. In living systems, diffusion of some substances would be slow or difficult without membrane proteins.
concentration gradient: an area of high concentration beyond from an expanse of low concentration
diffusion: a passive process of send of low-molecular weight material down its concentration gradient
facilitated send: a process by which material moves down a concentration gradient (from high to low concentration) using integral membrane proteins
hypertonic: describes a solution in which extracellular fluid has college osmolarity than the fluid inside the cell
hypotonic: describes a solution in which extracellular fluid has lower osmolarity than the fluid inside the cell
isotonic: describes a solution in which the extracellular fluid has the same osmolarity as the fluid within the cell
osmolarity: the full amount of substances dissolved in a specific corporeality of solution
osmosis: the transport of water through a semipermeable membrane from an area of high water concentration to an area of low water concentration across a membrane
passive send: a method of transporting material that does not crave energy
selectively permeable: the characteristic of a membrane that allows some substances through but not others
solute: a substance dissolved in another to course a solution
tonicity: the amount of solute in a solution.
Media Attributions
- Figure 3.24: modification of piece of work by Mariana Ruiz Villarreal
- Effigy 3.26: modification of work by Mariana Ruiz Villarreal
- Effigy 3.27: modification of work by Mariana Ruiz Villarreal
Source: https://opentextbc.ca/biology/chapter/3-5-passive-transport/
0 Response to "what cellular structure is selectively permeable for specific items to freely pass through it"
Publicar un comentario